Zirconium dioxide layers | ||||
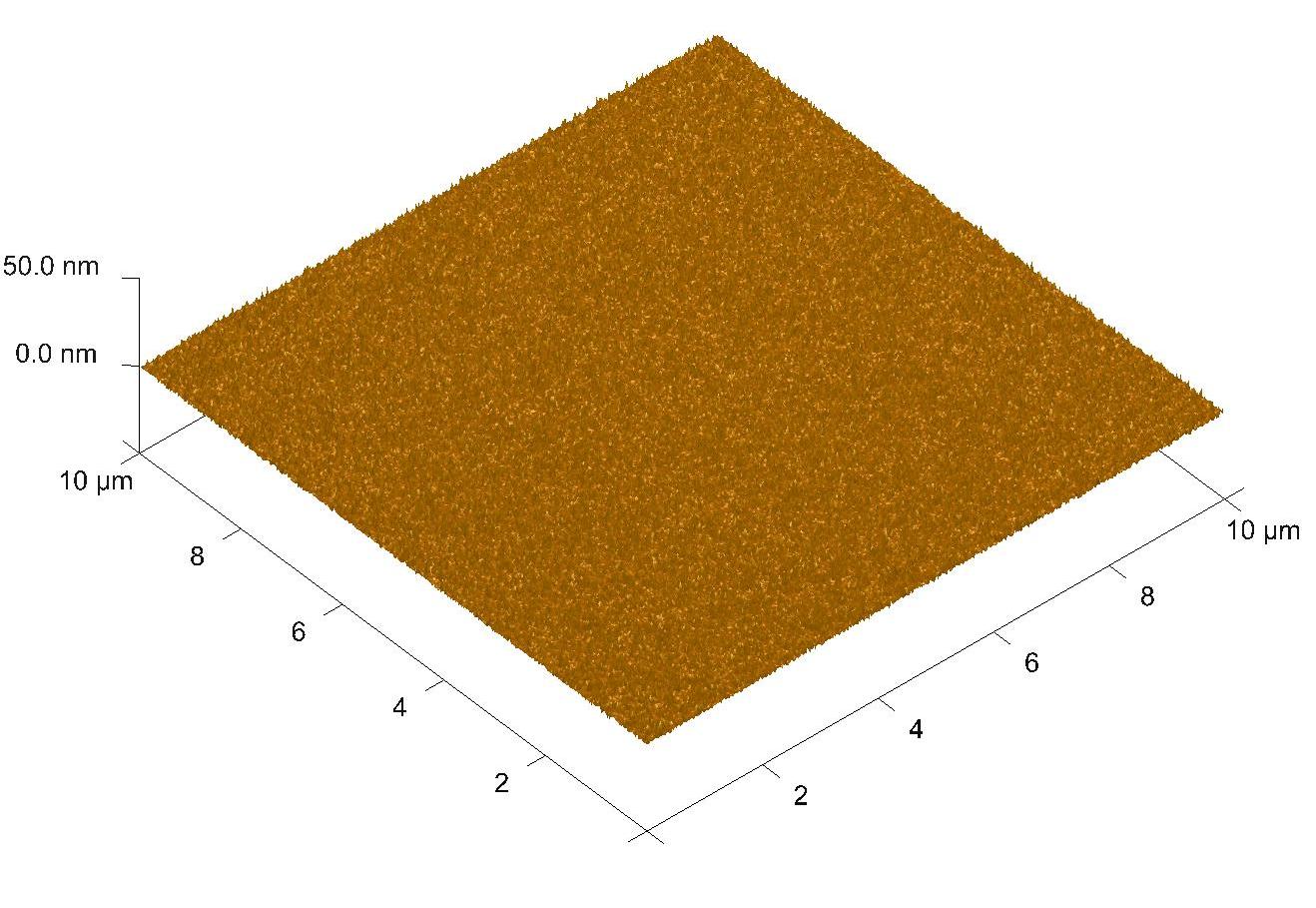
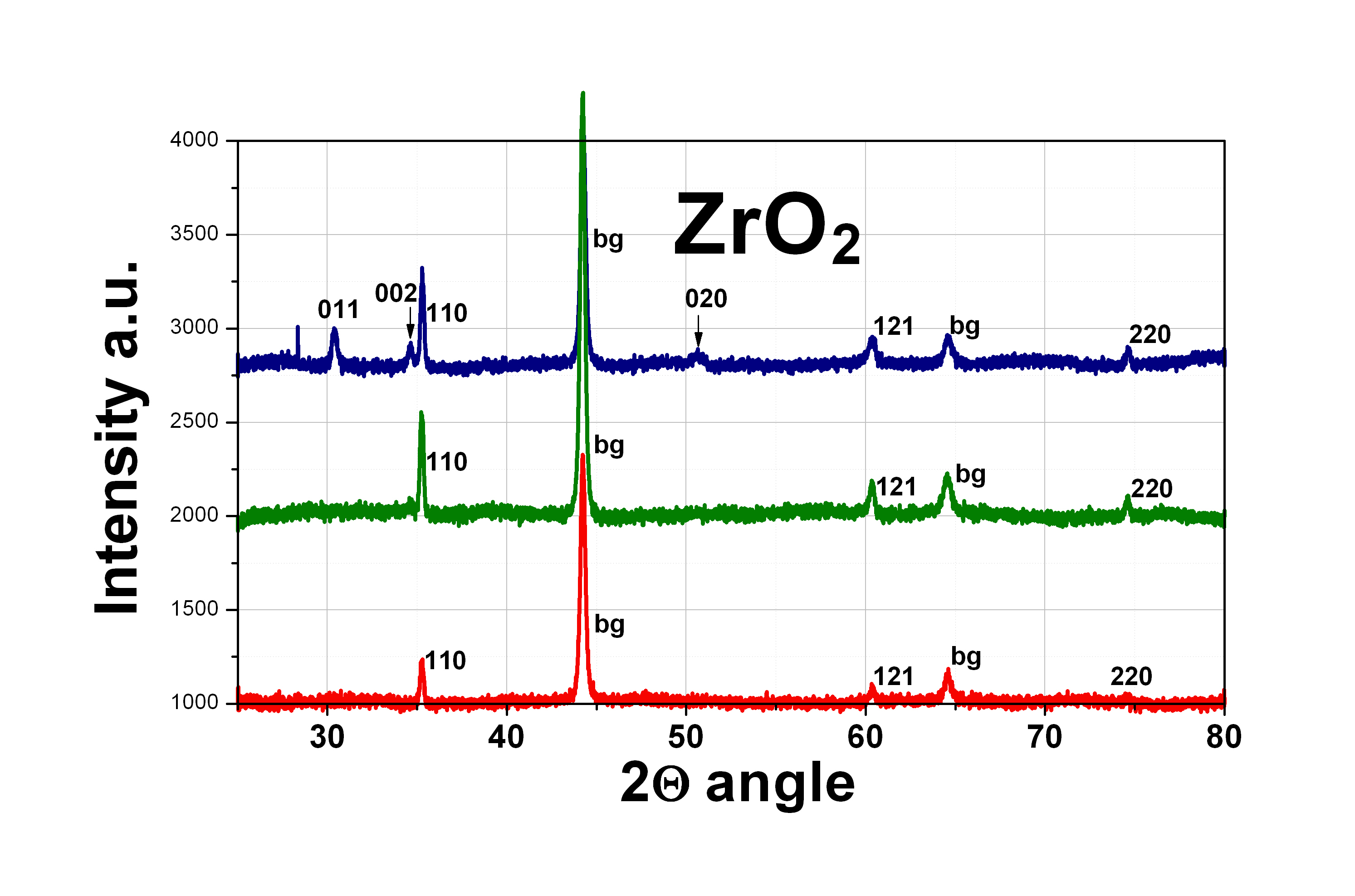
DescriptionZirconium dioxide, also known as zirconia (ZrO2), thin flims are obtained by atomic layer deposition (ALD) on range of substrates, like Si, GaN, SiC, graphene, SiO2 etc. The material is formed in a double exchange chemical reaction between two reactants (precursors) such as a deionized water (oxygen precursor) and a tetrakis(dimetyloamido)zirconium(IV) - TDMAZ (zirconium precursor). ZrO2 can be grown at the temperature range from 70°C to 300°C. The maximum size of the substrate is 20 cm of a diameter.
Specification
ApplicationsZrO2 is widely studied material because of the interesting physical and chemical properties. This material is characterized by high hardness and mechanical strength, good dielectric properties, low thermal conductivity, chemical stability and high refractive index. This oxide material is biocompatible, thus can find applications in medicine and dentistry for the manufacture of prostheses and implants. ZrO2 can be used as an insulator in electronic devices, as coating with high refractive index of optical elements in lasers and microscopes, and as barriers and protective layers as the active layer in photovoltaic structures. |
| |||